Is Vitamin C Acidic Or Alkaline
Open access peer-reviewed chapter
Vitamin C
By Nermin M. Yussif
Submitted: April 9th 2018 Reviewed: October 1st 2018 Published: December 5th 2018
DOI: 10.5772/intechopen.81783
Abstract
Vitamin C or ascorbic acid is one of the most common and essential vitamins. Due to its protective role, the supplementation of vitamin C becomes a must especially during the higher pollution levels. A day after day, scientists and researchers discover new functions for vitamin C. It was and still one of the cheapest treatment modalities that could preserve and protect human beings from infections, toxification, autoimmune diseases and cancer development. The role of vitamin C in providing better esthetics exhibits great importance. Its role as anti-aging agent preserves skin color and texture. Although it is not naturally synthesized in our bodies, our food is entirely rich of it.
Keywords
- vitamin C
- ascorbic acid
- chemical structure
- bioavailability
- functions
- different routes of administration and needed dosage
1. Introduction
In 1970, Pauling [1] stated that "Ascorbic acid is an essential food for human beings. People who receive no ascorbic acid (vitamin C) become sick and die."
Advertisement
2. Vitamin C source
Vitamin C is produced only in non-humans as primate species, guinea pigs, fishes and birds [2]. Although most of the animals have the ability to synthesis their needs of vitamin C, humans suffer from mutation in the DNA coding of gulonolactone oxidase which is the main enzyme responsible for ascorbic acid synthesis [3]. Due to this mutation, the external supplement of vitamin C becomes a must [4].
The main source of vitamin C for human beings is mainly found in fruits and vegetables. Citrus fruits and other types are particularly rich sources of vitamin C as; cantaloupe, water melon, berries, pineapple, strawberries, cherries, kiwi fruits, mangoes, and tomatoes. Furthermore, vegetables are considered the main source of vitamin C due to its higher content and availability for longer period throughout the year such as cabbage, broccoli, Brussels sprouts, bean sprouts, cauliflower, mustard greens, peppers, peas and potatoes [5].
Advertisement
3. Chemical structure of ascorbic acid
Although vitamin C is the generic name of l-ascorbic acid, it has many other chemical names as ascorbate and antiscorbutic vitamin. l-Ascorbic acid molecule is formed of asymmetrical six-carbon atoms (C6H8O6) which is structurally related to glucose (Figure 1) [7, 8]. Its molecular weight is 176 with a melting point of 190–192°C (with decomposition) and shows a density of approximately 1.65 g/cm3. l-Ascorbic acid is freely soluble in water (300 g/L at 20°C), difficult in alcohol (20 g/L at 20°C) and insoluble in chloroform, ether and benzene. It forms a clear colorless to slightly yellow solution. It has two p

Figure 1.
Chemical structure of vitamin C [
The chemical structure of ascorbic acid determines its physical and chemical properties. It is a weak, water soluble, unstable organic acid which can be easily oxidized or destroyed in light, aerobic condition (oxygen), high temperature, alkali, humidity, copper and heavy metals. Ascorbic acid is usually found in the form of white or slightly yellowish crystalline powder. Its crystalline form is chemically stable in dryness. However L-ascorbic acid is highly soluble in water, it shows great difficulty to be soluble in alcohol, chloroform, ether and benzene. In water, it forms clear colorless slightly yellow solution which is rapidly oxidized [8, 10].
There are many derivatives of ascorbic acid as sodium l-ascorbate (sodium ascorbate), calcium l-ascorbate (calcium ascorbate), zinc-ascorbate, 6-palmityl-l-ascorbic acid (ascorbyl palmitate) and ascorbyl monophosphate calcium sodium salt (sodium calcium ascorbyl phosphate) [128, 129].
Ascorbic acid is obtained from sodium ascorbate by cation exchange. While sodium ascorbate results from reacting methyl-d-sorbosonate (or ketogulonic acid methyl ester) with sodium carbonate. Calcium ascorbate is produced by the interaction of ascorbic acid with calcium carbonate in water and ethanol, which it is then isolated and dried. Ascorbyl palmitate is prepared by reaction of ascorbic acid with sulfuric acid followed by esterification with palmitic acid. Sodium calcium ascorbyl phosphate resulted from the reaction of ascorbic acid (alone or in combination with sodium ascorbate) with calcium hydroxide and sodium trimetaphosphate. The previous ascorbic acid derivatives have superior properties in comparison to ascorbic acid as the light resistance, skin irritation [128, 129].
Advertisement
4. Vitamin C metabolism
Vitamin C functions depend mainly on its main character as a reducing agent and the results of its oxidation mechanisms either reversible or irreversible [130, 131]. These reactions depend only on the pH changes and not on the presence of air or oxidizing agents [132].
Ascorbic acid undergoes a 3-step oxidation process. In the beginning, ascorbic acid can reversibly oxidize into dehydroascorbic acid on the exposure to copper, low alkaline media and heat [11].
Dehydroascorbic acid is a very short half-life (few minutes) product which can either reversibly or irreversibly oxidize in the tissues. In pH 4.0, ordinary temperatures and aqueous media, dehydroascorbic acid can be oxidized irreversibly into 2,3-diketo-l-glutonic acid (diketogulonic acid). However, the dehydroascorbic acid oxidation begins in mild acidic media (pH 4.0), it requires a neutral or alkaline media to progress more rapidly. The resultant diketogulonic acid is a stronger reducing agent, not reduced by glutathione or H2S and not an anti-ascorbutic agent. It was found that below pH 4.0, diketogulonic acid losses its reducing property. In acidic media and the presence of H2S, dehydroascorbic acid can also reversibly change into ascorbic acid. Ascorbic acid and dehydroascorbic acid have the same anti-ascorbutic effect [133, 134].
The third oxidation product is l-threonic acid and oxalic acid which proceed only in alkaline media (pH 7–9) [11]. All reversible changes can be done in the presence of H2S and glutathione in neutral or alkaline media. Sometimes, carbon dioxide may be the result of vitamin C oxidation at high doses [132].
In human beings, ascorbic acid is reversibly oxidized into dehydroascorbic acid, which can be reduced back to ascorbic acid or hydrolyzed to diketogulonic acid and then oxidized into oxalic acid, threonic acid, xylose, xylonic acid and lyxonic acid. Further oxidation (decomposition) may occur by the oxidizing agents in food. According to the oxidation-reduction reactions, ascorbic acid is the reduced form of vitamin C while dehydroascorbic acid is the oxidized form of vitamin C. The l-isomer of ascorbic acid is the only active form. Other isomers as d-ascorbic acid, d-isoascorbic acid and l-isoascorbic acid are present. These stereoisomers have no effect in the treatment of scurvy [128, 129].
The absorbed and the unabsorbed forms of ascorbic acid can be excreted in conjugated or non-conjugated pattern. Ascorbic acid may undergo limited conjugation with sulfate to form ascorbate-2-sulphate, which is excreted in the urine. Unchanged ascorbic acid and its metabolites are excreted in the urine. In the presence of intestinal flora, high doses of ascorbic acid (unabsorbed part) can oxidized into carbon dioxide which is the main excretory mechanism of vitamin C in guinea pigs, rats and rabbits. There exists equilibrium between ascorbic acid and dehydroascorbic acid, dependent on the redox status of the cells [12].
Advertisement
5. Vitamin C bioavailability
The bioavailability is a measure of the efficiency of gastrointestinal tract absorption [13].
5.1 Vitamin C absorption (active transport)
The hydrophilic nature of ascorbic acid facilitates its absorption through buccal mucosa, stomach and small intestine. Its absorption depends mainly on passive diffusion through the buccal mucosa [14].
Vitamin C absorption occurs through small intestine (distal intestine) by active transport mechanism. Sodium electrochemical gradient is the process by which active transport of ascorbic acid occurs. This process proceeds by the help of sodium vitamin C transporter type 1 (SVCT1). This is the same transporter responsible for vitamin C transport in retina. SVCT2 is responsible for transporting vitamin C into brain, lung, liver, heart and skeletal muscles [15]. The absorption process is usually inhibited by glucose [135].
The majority of ascorbate is transported by SVCT1 in epithelial cells (e.g., intestine, kidney and liver), and the remaining is transported by SVCT2 in specialized cells (e.g., brain and eye) [15, 16]. The main concentrations of vitamin C are located in brain and adrenal cells.
The oxidative products of vitamin C as dehydroascorbic acid are transported faster into cells than the pure form [17].
While the absorption of low doses (15–30 mg) is very high (up to 98%), ascorbic acid absorption decreases (50%) with larger doses (1000–1250 mg) which is commonly administrated in acute illness [136, 137].
In human blood, ascorbic acid is always found in the reduced form (ascorbic acid). It was also found that the red blood corpuscles are not permeable for ascorbic acid and also to glucose. It oxidized very slowly in blood than in plasma (no oxidation reactions occur) [132]. Its normal plasma level ranges between 50 and 100 μM according to the diet intake in healthy non-smoker individuals [137]. Increasing the plasma level and the intracellular level is not a dose dependent. Its intracellular level is higher than the plasma level. The plasma level does not increase above the normal range even by increasing the intake into 500 mg because of its excellent excretion from kidneys through urine [13].
5.2 Vitamin C distribution
Vitamin C is widely distributed in all the body tissues. Its level is high in adrenal gland, pituitary gland, and retina. Its level decreases in kidneys and muscles.
5.3 Vitamin C excretion
Vitamin C metabolites (oxalate salts) and unmetabolized vitamin C are excreted by kidneys. Few percentage of vitamin C is excreted through feces.
The urinary excretion of vitamin C is dose dependent. Less than 100 mg/day, no vitamin C was detected in urine. At 100 mg/day, 25% of its amount was excreted in urine. The latter percentage is doubled with the administration of 200 mg/day [13, 18].
At high doses, large amount of unmetabolized vitamin C is excreted. The higher doses of vitamin C intake, the higher vitamin C concentration in blood and tissues occurs. As a response for high doses, vitamin C excretion from kidneys and sweat occur. The antiviral and anti-bacterial effect of vitamin C protects skin and kidneys from infection [1]. Also in extra doses, the oxidation components were used as an anticancer effect more the vitamin C itself [138].
It was found that the excretion of ascorbic acid when administrating 400 mg ascorbic acid ranges between 30 and 50% in healthy individuals. This percent decreases in diseased patients due to higher consumption. Repeated low doses (about 200 mg) are highly recommended in diseased individuals due to theses low doses saturate the body. Extremely low dosages (90 mg/day) could result in inability of the immune system to respond to diseases as degenerative diseases. Therefore, limited renal clearance of ascorbic acid is usually detected. The plasma saturation of ascorbic acid at 70 μM (0.123 mg/dl). This level controls the excretion of the ascorbic acid through kidneys. At Higher plasma levels (above 70 μM), higher excretion levels are usually detected. The intravenous route exerts 30–70 folds of vitamin C plasma levels than the oral route [19].
The rapid excretion due to its water soluble nature limits its harmful effect and makes it totally safe product in normal doses. It also found that the upper tolerable limit (UL) is 2 g. Depending on the depletion-repletion study, it was found that the RDA is 75 mg for women and 90 mg for men. It was modified by Levine et al. in 2001 into the administration of 90 mg to both sexes. The maximum bioavailability and absorption of vitamin C achieved at 500 mg [20].
5.4 Vitamin C storage
In 1936, Marinesco et al. detected the lower levels of ascorbic acid in others organs as pancreas, spleen and thymus gland. Plaut and Billow detected the ascorbic acid lowering not only in the organs but also in body fluids as CSF, blood and urine. They also detected this deficiency in neural diseases and alcoholism. Many reasons were thought to be the cause of vitamin C deficiency in old people. Decreased intestinal absorption and dietary deficiency are the main causes.
In human blood, ascorbic acid is always found in the reduced form (ascorbic acid). It was also found that the red blood corpuscles are not permeable for ascorbic acid and also to glucose. It oxidized very slowly in blood than in plasma (no oxidation reactions occur) [132]. Its normal plasma level ranges between 50 and 100 μM according to the diet intake in healthy non-smoker individuals [137]. Increasing the plasma level and the intracellular level is not a dose dependent. Its intracellular level is higher than the plasma level. The plasma level does not increase above the normal range even by increasing the intake into 500 mg because of its excellent excretion from kidneys through urine [13]. In 1934, Yavorsky et al. [21] analyzed the ascorbic acid amount found in the different body organs at different ages (Table 1).
| 1–30 days (g) | 11–45 years (g) | 46–77 years (g) | |
|---|---|---|---|
| Adrenals | 0.581 | 0.393 | 0.230 |
| Brain | 0.460 | — | 0.110 |
| Liver | 0.149 | 0.135 | 0.064 |
| Kidney | 0.153 | 0.098 | 0.047 |
| Lung | 0.126 | 0.065 | 0.045 |
| Heart | 0.076 | 0.042 | 0.021 |
Table 1.
Vitamin C storage in different organs at different ages.
Advertisement
6. Vitamin C functions
6.1 Role in immune system and inflammation
Vitamin C has an important role in the maintenance of a healthy immune system and its deficiency causes immune insufficiency and multiple infections. The ascorbic acid level is lowered in various body fluids during bacterial infections. Thus, it is commonly used as adjunctive treatment in many infectious diseases such as hepatitis, HIV, influenza and periodontal diseases [22].
Vitamin C administration modifies and enhances both the innate and adaptive immune response. It neutralizes the bacterial toxins especially endotoxins by blocking the essential signal for lipopolysaccharides (LPS) formation. On the other hand, LPS block the passage of ascorbic acid through blood brain barrier and inhibits its uptake by various cells [22].
Ascorbic acid improves the phagocytic properties and activity of various immune cells including neutrophils, natural killer cells, macrophages and lymphocytes. Vitamin C increases lymphocytes proliferation and antibody production [23, 24].
6.2 Anti-oxidant property
Oxidative stress/ROS have a main role in inflammatory diseases including periodontal diseases [25]. The ROS are classified into 3 classes; the first are reactive free radicals as oxygen related radicals (superoxide, hydroxyl radical or peroxyl radicals). The second class is reactive species but not free radicals as hypochlorous acid. The third class is radicals resulted from the reaction with ascorbic acid [26]. Antioxidants are also classified into enzymatic and non-enzymatic. The enzymatic antioxidants include catalase enzyme, thiol-containing agents (cysteine, methionine, taurine), glutathione and lipoic acid [27].
Vitamin C is one of the nutrient non-enzymatic anti-oxidants [28, 29, 30]. Its antioxidant effect is by electron donation process where vitamin C easily donates two electrons (reduction reaction) to other compounds in order to prevent its oxidation. When ascorbic acid donates the first electron, it is transformed into a free radical called ascorbyl radical (semi-dehydroascorbic acid). It is a relatively stable, unreactive free radical with unbound electron in its outer shell but it has a short life time (10–15 s). The unreactivity of this radical makes it unharmful to the surrounding cells. This process is called free radical scavenging or quenching. When it donates the second electron, it transformed into dehydroascorbic acid. Its stability may only last for few minutes [28, 31].
As a general rule, it was detected that vitamin C acts as a pro-oxidant at low doses and acts as an antioxidant in high doses. It was also detected that the level of vitamin C in the skin usually exposed to ultraviolet radiation is lower than that exposed lesser.
The antioxidant activity of vitamin C enhances the epidermal turn over, and the movement of young cells to the surface of the skin where they replace old cells [32]. The study conducted by Frank in [139] showed that RNA improved the ability of the skin cells to utilize oxygen.
Ascorbyl radical and dehydroascorbic acid are reversible agents which can easily rebound into ascorbic acid. These reversible agents can irreversibly transformed into 2,3-diketogulonic acid which is further metabolized into xylose, xylonate, lyxonate and oxalate (Figure 2) [34].
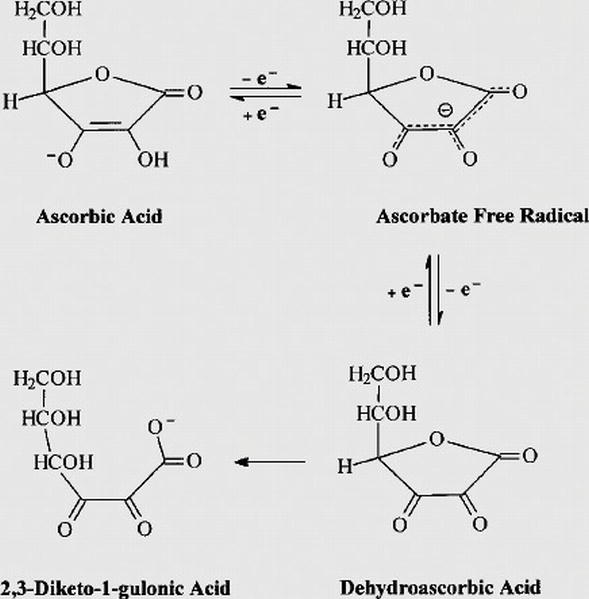
Figure 2.
Redox metabolism of ascorbic acid [
Vitamin C is considered as a strong anti-inflammatory agent as it inhibits many types of inflammatory mediators as tumor necrosis factor alpha [35]. This property is commonly used in the treatment of postoperative erythema formed after CO2 laser in skin resurfacing [36]. In 1987, Halliwell [37] detected significant reduction of plasma levels of ascorbic acid in association with elevated histamine in inflammatory diseases as ulcerative colitis and rheumatoid arthritis. This was explained by the discovery of the anti-histaminic effect of vitamin C. It was also found that the higher ascorbic acid content in joints, the higher protection levels against damage which directed many physicians to use ascorbic acid in combination therapy with drugs aiming to joint protection as glucosamine [37, 38].
It was discovered that vitamin C has an efficient chemotherapeutic effect. The cytotoxic effect of vitamin C is dose and route dependent. The tumor cells are more sensitive to high intravenous (cytotoxic) levels of vitamin C than the normal ones [140]. At the administration of 10 g of intravenous vitamin C, a marked elevation of the extracellular concentration (1000 μmol/L) is detected which have a toxic effect on the cancer cells due to the action of the ascorbyl radicals (free radical species) [39]. On the contrary to the cancer cells, normal cells can compensate the damage occurred by these oxidative species [141]. It was also found that these mega doses of vitamin C are essential in other diseases as diabetes, cataracts, glaucoma, macular degeneration, atherosclerosis, stroke and heart diseases [40].
Vitamin C improves the immune system and its deficiency causes immune insufficiency and multiple infections. It was found that vitamin C modifies the behavior and activity of the immune cells; it also improves the phagocytic properties of neutrophils and macrophages. In addition, vitamin C increases the antibody production, concentration of antibodies and the activity of lymphocytes [41]. It was detected that the level of vitamin C in leukocytes is higher than its level in plasma because they have the ability to store it [142].
Vitamin C is commonly used as an adjunctive treatment in many infectious diseases as hepatitis, HIV, common cold and influenza. It has an important role in the antibacterial reactions performed in our body by neutralization of the bacterial toxins especially endotoxins [42]. It was found that 100 μM/L ascorbic acid can lower the bacterial replication (bacteriostatic effect) [143].
Sufficient amount of vitamin C causes blockage of the signaling essential for lipopolysaccharides (LPS) formation. It also stops the production of ROS especially reactive nitrogen species which is mainly produced during infection [42]. In bacterial infections, the level of ascorbic acid in various body fluids is lower than usual which perform further depression due to the action of LPS in blocking the passage of ascorbic acid through blood brain barrier. LPS also inhibits the uptake of various cells to ascorbic acid [143].
The anti-aging effect of vitamin C is regarding to its potent antioxidant effect, its stimulatory effect of enhancing the collagen formation, protection of the persistent collagen especially elastin against damage and finally, inhibits the cross-linking effect formed in wrinkles [144]. It was found that the amount of ascorbic acid changes with age. The younger the age, the higher the ascorbic acid level present. In 1934, Yavorsky et al. [21] analyzed the ascorbic acid amount found in the different body organs at different ages.
6.3 Depigmenting effect
It was found that the higher the ROS is, the deeper the pigmentation produced. Anti-oxidants act a great role in lowering the melanin formation [43]. Vitamin C is considered a potent depigmenting agent which is used in the treatment of various cases of skin hyperpigmentation [44, 45, 46, 47, 48].
It can be used as an adjunctive treatment in melasma and severe cases of hyperpigmentation and as a treatment in mild and moderate cases [49].
Vitamin C inhibits melanogenesis in different steps via more than one mechanism [145]. Being an anti-oxidant, ascorbic acid prevents production of free radicals which triggers melanogenesis [50]. It reduces o-dopaquinone back to dopa, preventing dopachrome of 5,6-DHICA [51] and reduces oxidized melanin changing the pigmentation from jet black to light tan [52]. Furthermore, the direct suppression of tyrosinase enzyme exhibits a great property [53].
It can be used as an adjunctive treatment in melasma and severe cases of hyperpigmentation and as a treatment in mild and moderate cases [49]. The higher the ROS is, the deeper the pigmentation produced. Antioxidants act a great role in lowering the melanin formation [43].
Other mechanisms of blocking melanogenesis include inhibition of tyrosinase activity by interacting with copper ions at active sites of the enzyme [51, 53]. In addition, vitamin C inhibits melanocyte proliferating agents (IL-1, MSH, and PGE2) and peroxidase reactions on melanocytes [54].
In 1970, Pauling and Cameron [146] discovered that vitamin C has an efficient chemotherapeutic effect. The cytotoxic effect of vitamin C is due to the action of the ascorbyl free radical and it is dose as well as route dependent [55]. The tumor cells are more sensitive to high intravenous (cytotoxic) levels of vitamin C than the normal ones [23, 56]. A synergistic effect is detected between the intravenous vitamin C administrations accompanying the tumor cytotoxic agent in patients suffering from advanced cancer [57].
Melanoma is the most commonly treated malignant tumors using vitamin C due to the high susceptibility and sensitivity of its cells to vitamin C. It induces sodium ascorbate induced apoptosis of melisma. The lethal effect of ascorbic acid is attributed to inhibiting the production of IL-18 (essential for melanoma proliferation), change the intracellular iron level [56, 58].
Besides the antioxidant role, ascorbic acid also acts as an electron donor for eight enzymes. Three of these enzymes are involved in collagen formation [34]. Other two enzymes are responsible for carnitine formation, one enzyme is responsible for epinephrine production from dopamine, and the other is responsible for the addition of the amide groups into peptide hormones. Finally, it is essential for tyrosine metabolism and melanin production. The anti-tyrosinase enzyme occurs at 0.05–0.50 mM intracellularly [31].
6.4 Collagen production
The role of vitamin C in collagen formation is well known. Vitamin C is an essential factor for the hydroxylation of proline, cofactor during collagen processing, activation of pro-collagen messenger RNA, inhibition of matrix metalloproteinases (MMPs) that are responsible for collagen fibers degradation and fibroblast activation intended for new and proper collagen formation [12, 59, 60, 61].
As regards the effect of ascorbic acid on periodontal ligament, it enhances the periodontal ligament maturation and renewal by induction of the collagen formation especially collagen III (young collagen) and keeps the balance between collagen I (mature collagen) and III for tissue maturation. It was detected that thicker periodontal ligament were detected near the CEJ and narrower ones were detected in the middle one third of the root due to the effect of vitamin C in keeping the collagen bundles, well organized and more resistant to tension. Furthermore, it also activates the fibroblast itself; proliferation, production and differentiation. By the vitamin C role in modifying the produced collagen type IV through its role as a cofactor in hydroxyproline synthesis and improving the endothelial cell vitality, its role in angiogenesis could not be forgotten. In periodontal disease, it is recommended to use an adequate well calculated dosage of vitamin C to achieve higher level of healing, minimal bleeding, higher quality of the newly formed tissues and increasing the resistance of tissues to future destruction [12, 62].
Collagen is the main component of bone matrix. Many publications had confirmed the role of vitamin C in bone formation. In postmenopausal women, higher levels of vitamin C are needed in order to reduce the incidence of osteoporosis [63, 64].
When vitamin C is used with scaffolds in tissue engineering, the sustained release of vitamin C stimulates the formation of type I collagen and alkaline phosphatase enzyme [65].
Vitamin C is a potent factor in the extracellular bone matrix proteins formation as collagen type I, osteonectin and osteocalcin. Its combination with vitamin E has an essential role in the proliferation and differentiation of the osteoblasts [66].
The amount of ascorbic acid in human body changes with age. The younger the age is, the higher the ascorbic acid level present [67].
Vitamin C enhances the collagen formation (collagen type I) and protects the persistent collagen to resist damage. Finally, it inhibits the cross-linking effect found in wrinkles [68].
The anti-oxidant property is also involved in the anti-aging effect; vitamin C plays an important role in protecting the cellular integrity as it scavenges the ROS, prevents oxidation of the cellular proteins, lipids as well as DNA and protects the cellular junctions. It also improves the tissue vasculature [69].
As regards the effect of ascorbic acid on periodontal ligament, it enhances the periodontal ligament maturation and renewal by induction of the collagen formation especially collagen III (young collagen) and keeps the balance between collagen I (mature collagen) and III for tissue maturation. Thicker periodontal ligament were detected near the cement-enamel junction (CEJ) and narrower ones were detected in the middle one third of the root due to the effect of vitamin C in keeping the collagen bundles, well organized and more resistant to tension. It also modifies the rate of fibroblast proliferation [12, 62, 70] (Figure 3).

Figure 3.
(a) Human fibroblast culture and (b) human fibroblast proliferation after treated with vitamin C [
6.5 Others functions
6.5.1 Vitamin C and metal absorption
Vitamin C increases the absorption of heavy metals from the intestine as iron. Vitamin C has an important role in the carnitine synthesis which is an enzyme co-factor that increases the absorption of non-haem iron in GIT. It also enhances production of reduced iron which is the preferred form for the intestinal absorption [72]. It was found that factory workers have high oxidative stress. The cause behind the latter observation was the high plasma levels of lead (73 μg of lead/dl), thiobarbituric acid (46.2%) and chloramphenicol acetyltransferase (70.3%). By the administration of combination dosage of vitamin C (1 g) and vitamin E (400 IU), great improvement of the general health with the plasma levels returned to its non-lead levels [73].
6.5.2 Vitamin C and drug absorption
Combination of vitamin C supplements with aspirin and opiates has a strong synergistic effect on these drugs [54, 74]. On the other hand, oral contraceptive pills increases ascorbic acid turnover and reduce level of ascorbic acid [75].
In addition, many vitamins including vitamin E (α-tocopherol), vitamin B15 (carnitine), tryptophan and folic acid require vitamin C for their absorption [76]. The combination of vitamin C and E enhance the efficiency and life span of vitamin E by providing sustained release effect and regeneration of the oxidized vitamin E [59, 77]. Thus, vitamin C deficiency is considered a cause of some of these vitamins deficiency such as folic acid deficiency [78, 79, 80].
Furthermore, marked increase of ascorbic acid turnover was reported on consumption of estrogen containing medications [81].
As a result of oxidation reaction, the production of hydrogen peroxide is enhanced by cations as iron and copper [19].
EDTA is the only permissible preservative that could be used with injectable vitamin C products [82].
Limited evidence suggests that ascorbic acid may influence the intensity and duration of action of bishydroxycoumarin.
6.5.3 Vitamin C and lipid metabolism
Although vitamin C is water soluble, it has great effects on the lipids either intracellularly or extracellularly. Vitamin C is an essential factor protecting the lipids of cell membrane from oxidation. It also protects the lipid bilayer of skin.
Significant control of hypertension and high cholesterol levels [147]. It was observed that vitamin C improves the lipid metabolism by inhibiting the oxidation of the unsaturated lipids and lipoproteins (scavenging effect) [83]. ROS can easily oxidize the cellular lipoproteins and the circulatory LDL and results in lipid peroxidation. By a process called radical propagation, the formed lipid peroxides can easily reacts with oxygen to finally form lipid hydroperoxides. Ascorbic acid can easily inhibit this process by reducing the ROS [84]. It was found that vitamin C decreases the oxidation of lipids and in lowering the low density lipoprotein (LDL) cholesterol [85].
Vitamin C is a cofactor in many processes in various organs including catecholamine biosynthesis and steroidogenesis as well as lowering cholesterol and bile acids [86, 87, 88]. However, vitamin C deficiency is linked to many diseases and disorders such as fatty liver [89], hyperlipidemia [90], obesity and diabetes mellitus type II [91].
Vitamin C has an important role in decreasing the atherosclerosis and the inhibiting the thrombus formation through decreasing the platelet aggregation [148].
6.5.4 Vitamin C and bone formation
As previously mentioned, vitamin C increases the production of collagen type I and X needed for matrix formation, activation of osteoblast growth and differentiation [92]. It is also needed in order to maintain adequate bone density [93, 149]. Stimulation and higher expression of osteocalcin and osteonectin on the osteoblasts was also reported [66].
In postmenopausal women, higher levels of vitamin C are needed in order to protect against bone abnormalities as it is considered as delaying osteoporosis factor [149]. Furthermore, in scurvy, lower bone density with marked bone abnormalities commonly reported [94, 95]. In case of deficiency occurs in young individuals, bone fragility, cartilage resorption and fracture of growth plates. The detected abnormalities were attributed to reduced activity of osteocytes and chondrocytes [93, 96]. It also maintains and preserves the balance between osteoblasts and osteoclasts [97].
In order to achieve optimum proliferation of the osteoblasts and fibroblasts, 200 μg/ml is the maximum dose needed. Apoptosis occurs when exceeding such dose [39, 98].
In 2004, an in vitro study used vitamin C with scaffolds in tissue engineering in order to regenerate bone. The sustained released vitamin C stimulates the formation of type I collagen and alkaline phosphatase [65].
In 2013, Fu et al. [150] used isotonic irrigation of ascorbic acid derivative during grafting of the anterior tendon of the knee joint. Significant reduction of the inflammatory response in the surgical site due to lack of toxicity, irritation, watery consistency and potent anti-oxidant effect.
6.5.5 Vitamin C and stress (cortisone)
Vitamin C has an important role in controlling the stress [41]. In stress, the overproduction of cortisone affects the defense mechanisms [151]. This was explained by the reduction of glucose levels [152].
6.5.6 Anti-microbial properties
Although the toxic effect of high doses, it shows great benefits on the other hand. Vitamin C excretion depends mainly on kidneys and sweat. The antiviral and anti-bacterial effect of vitamin C protects skin and kidneys from infection [1]. Also in extra doses, the oxidation components were used as an anticancer effect more the vitamin C itself as mentioned before [138].
Advertisement
7. Vitamin C dosage
The dilemma behind vitamin C dosage has been started many years ago. Dosage calculations differ according to the medical status, aim of administration either prophylactic or curative, route of administration and patient age.
When we talk about ascorbic acid average dosage, we have to differentiate between four terms; the estimated average requirement (EAR), adequate intake level, the tolerable upper intake level (ULs) and the recommended dietary allowance (RDA). The EAR is used to calculate the RDA and the adequate intake levels. While the UL is the level below which toxic effects have not been seen [99].
Because of the poor oral bioavailability of vitamin C, toxic signs and symptoms may appear with large doses exceeding 1000 mg or utilizing more than 2 g as single dose [100].
7.1 Dosage for healthy individuals
For healthy individuals, vitamin C dosage differs according to age and sex. The average normal content of ascorbic acid in human body is about 1.5 g. Daily, our body usually consumes 3–4% (40–60 mg) of this pool. In order to keep this pool balanced, the daily oral intake should be 200–300 mg of vitamin C. The daily intake of 5 fruits or vegetables will provide a 200 mg of vitamin C which will result in 70 μM plasma level. It was also found that the uptake of vitamin C differs from tissue to another [13, 31].
As an average, the RDA of vitamin C for adult healthy men is about 90 mg and for healthy women is about 75 mg. The ULs dosage was calculated at a level of 2 g per day for healthy individuals [99, 100]. Based on gender, the RDA of both males and females should not be less than 90 mg which secure the neutrophil saturation and urinary excretion. The maximum bioavailability could be reached at 500 mg dose [155]. It was found that the oral route could produce a maximum 220 μmol/L of vitamin C plasma concentration even with high dosage administration while the intravenous route produces higher levels which could reach to 1760 μmol/L [47].
The recommended dosage also differs according to the country or health institute that recommended this dosage. The recommended dosage of vitamin C can only maintain its plasma level constant up to 5–6 h only [57].
Another suggestion said that the recommended dosage of vitamin C is 100–120 mg/day. It was discovered that dose elevation could not control developing of related diseases (Gomez-Romero et al., 2007). In 2010, the Japanese ministry of health recommended 100 mg for both men and women [153]. Older researches recommend the average suitable doses for vitamin C is 100–3000 mg/day. In 1999, Levine et al. [101] determinate the average daily range of vitamin C is 210–280 mg only by food.
The bowel tolerance dose is the dose just below which produces diarrhea. The bowel tolerance usually differs from one to another according to the medical status. It can be determined only by trail. High dosage ranging between 3 and 6 g is recommended till diarrhea occurred. The dosage then has to be decreased till the bowel balance achieved again. Such side effect (diarrhea) is useful in treatment in patients with constipation [102]. Vitamin C is well absorbed up to 500 mg/day [103].
7.2 Dosage for smokers
A higher dose of vitamin C is strongly recommended in order to compensate for the smoking hazards and neutralize for the resulted oxidative stress [104]. In 2000, Sargeant et al. [105] reported the protective effect of vitamin C administration in smokers against obstructive airway disease.
A 110 mg is the recommended daily dosage for smokers due to its antioxidant level is below normal. The lower plasma level of ant-oxidants regards to their lower consumption healthy food, higher levels of toxic products which produce oxidative stress [106, 107].
7.3 Dosage in surgery and illness
Vitamin C is an important partner in the parental nutrition especially in acute conditions (shocked surgical, trauma, burn and septic conditions) [99]. In surgery and illness, redistribution and reabsorption of vitamin C occurs. It can be assumed that increased demand in tissue results in a decrease in its plasma concentration. It was discovered that patients can tolerate much more doses of vitamin C than healthy individuals [22, 102, 108].
During acute inflammatory phase as postsurgical, trauma, sepsis, and burns, the plasma level of vitamin C significantly lowered [154]. After surgery, the blood concentrations of vitamin C decreased immediately and 7 days postoperative (44.3–17.0 mmol/ml). Also the urinary excretion of vitamin C is lowered in the first week postoperatively (3.12 and 1.94 mg/day) [155]. In severe burns, Tanaka et al. [109] used 95 g/day of vitamin C for patient weight of 60 kg in the first 24 h. Lowering the tissue edema is observed on vitamin C administration.
A 3 mg orally or 100 mg parentally is recommended in acute conditions to compensate the diseased conditions and maintain the physiologic functions [99].
In 2010, Fukushima and Yamazaki reported the intense decrease of the plasma vitamin C level in surgical complicated patients, especially surgical intensive care unit patients which leads to increase of the oxidative stress. It was found that the recommended daily doses of vitamin C (RDA) either orally or parentally are not sufficient. They recommended higher doses of vitamin C in order to compensate the large amounts consumed during illness. A 500 mg of parental vitamin C was recommended in uncomplicated surgical cases and higher doses were recommended in surgical intensive care unit patients.
In 2013, Hemilä [108] detected the significant reduction of the common cold induced asthma and respiratory infections incidence as a direct effect of oral or intravenous vitamin C in susceptible individuals.
7.4 Vitamin C dosage in old patients
In old aged individuals, vitamin C deficiency is a common feature. It is mainly attributed to improper intake within diet which is usually lower than the average recommended dietary allowance (RDA) by 25.9–38.3% [110, 111].
7.5 Vitamin C dosage in pregnant patients
In females, the hormonal changes result in increased oxidative stress. Lower vitamin C levels are usually detected in pregnant women due to several factors; obesity and iron intake which could result in low birth weight [112, 113, 114].
Up till now, there is no evidence supporting harmful effect on using ascorbic acid injection for pregnant females. Caution should be taken when administrating injection in nursing women.
7.6 Vitamin C dosage in cancer patients
The tumor cells are more sensitive to high intravenous (cytotoxic) levels of vitamin C than the normal ones [140]. At the administration of 10 g of intravenous vitamin C, a marked elevation of the extracellular concentration (1000 μmol/L) is detected which have a toxic effect on the cancer cells due to the action of the ascorbyl radicals (free radical species) [39]. On the contrary to the cancer cells, normal cells can compensate the damage occurred by these oxidative species [141]. It was also found that these mega doses of vitamin C are essential in other diseases as diabetes, cataracts, glaucoma, macular degeneration, atherosclerosis, stroke and heart diseases [40].
7.7 Vitamin C dosage in stressful patients
In people under stressful conditions either physically or emotionally, vitamin C deficiency is a common incidence. Serum levels of stress indicators were measured in pilots and reported as following; 21.1% higher malondialdehyde (MDA), 21.7% higher superoxide dismutase (SOD), and 25.1% higher total antioxidant capacity (TAC). Higher doses of vitamin C are recommended [156]. Among animals, their bodies can produce 5 times of vitamin C when exposed to stressful conditions. Doses of 30–200 times greater than the RDA of 90 mg/day are recommended during stress [157].
Advertisement
8. Vitamin C route of administration
Ascorbic acid is a water soluble vitamin which facilitates its absorption from buccal mucosa, gingival tissues, stomach and small intestine [14]. The literature reported several routes of vitamin C administration. In order to explain how we can reach the optimal route of administration and dosage, accurate analysis of the treated tissue condition and its nature in healthy and different pathological conditions is highly recommended.
By all routes of administration, the plasma level of vitamin C returns to its normal range within 24 h [31].
8.1 Oral route
The oral route of administration is the most common one which can be available either in the form of tablets, powder or solution. It is an essential element in multivitamins supplements [7, 31, 47].
8.2 Parental route
The intravenous route is used in advanced cancer therapy and severe illness as a complementary treatment [144]. On the contrary to oral route, the vitamin C plasma level cannot be controlled when administrated parentally. It was detected that the vitamin C plasma level is 25 fold higher than the level recorded by the oral doses [115]. It was detected that the vitamin C plasma level is 30–70 fold which is higher than the level recorded by the oral doses [116]. The urinary excretion levels were also elevated [47].
The intravenous vitamin C administration is usually used to treat hyperpigmentation especially in patients under chronic hemodialysis [47].
8.3 Topical route and topical formulations
The oral route not actually provides a source of vitamin C to peripheral structures as skin. It was detected that the vitamin C level in skin is very low. The only route that can provide a vitamin C source for skin is the topical or local routes. It was found that the usage of local application promotes the surgical healing and better tissue reconstruction [35].
Although the advantages of the topical route, the epidermal absorption of vitamin C is still limited. The water soluble nature of ascorbic acid is the main cause behind its limited penetration [60, 117, 118]. Ascorbyl palmitate and magnesium ascorbyl phosphate (buffered forms) are esters which provide higher lipid solubility through stratum corneum and higher photostability [117]. In 2002, Joung and Yi detected the low penetration power of the water soluble L-ascorbic acid into the lipophilic stratum corneum.
Acidity and concentration of topical vitamin C control its absorption. For optimal percutaneous absorption, acidic pH < 3.5 is required which is lower than the l-ascorbic acid pH (4.2) [60, 119]. Concentration of ≤20% is associated with high absorption and tissue saturation [119].
When dealing with oral mucosal tissues, vitamin C is easily absorbed by passive diffusion through the buccal mucosa [14]. It was found that the absorption of the ascorbic acid through the buccal mucosa and small intestine is nearly equal [120].
8.4 Transdermal route
Transdermal route is a big title enrolled many different techniques as sonophoresis and nanoparticles route that enhance the absorption and penetration of the topically delivered hydrophilic drugs. It can be used with insulin, morphine, caffeine, glucose, lidocaine and ascorbic acid. The micro-vibrations produced by using the ultrasound waves are the main responsible to increase the kinetic energy of the drug and deliver it deeper through the skin layers [121, 158].
8.4.1 Sonophoresis
It is one of the transdermal routes which used to increase the absorption and penetration of the topically delivered hydrophilic drugs. It can be used with insulin, morphine, caffeine, glucose, lidocaine and ascorbic acid. The micro-vibrations produced by using the ultrasound waves are the main responsible to increase the kinetic energy of the drug and deliver it deeper through the skin layers. The local and topical applications of drugs has many benefits in targeting the drug benefits directly to the area of interest and also the avoidance of the absorption, metabolism, excretion and dose problems.
In 2003, Huh et al. [45] studied the effect of iontophoresis and topical vitamin C in melasma treatment. Vitamin C solution and iontophoresis were applied. For better introduction, a low molecular weight solution was used instead of gel or cream. Significant reduction of pigmentation was detected after application.
8.4.2 Nanoparticles
l-Ascorbic acid is unstable formula of vitamin C when exposed to air, moisture, oxygen and base. The end products of l-ascorbic acid are 2,3-diketo-l-gulonic acid, oxalic acid, l-threonic acid, l-xylonic acid and l-lyxonic acid. Many researches tried to overcome this problem either by using its salts as magnesium ascorbyl phosphate or to encapsulate it using liposome, microemulsions, lipid crystals or inorganic component as hydrated ZnO. This encapsulation not interferes with the efficiency of the drug used but it controls its release and increase its penetration power [159].
8.4.3 Injection
In 2004, Senturk et al. [122] intraperitoneal injection of vitamin C was applied. Skin and serum specimens were taken at the end of the experiment. Significant reduction in the level of serum of inflammatory cytokines, IL-6 and TNF-alpha was detected. Improvement of tissue collagen was also detected.
Approximately 4 days are the detected half-life of the topically applied vitamin C (the remaining amount in tissues) [119].
Advertisement
9. Dosage calculation or different routes of administration
There is strong correlation between the applied dosages and the intestinal absorption rate. Marked absorption (98%) is reported in lower doses (15–30 mg). on the other hand, vitamin C absorption could be reduced to 50% in large doses that exceeds 1000 mg [15, 136, 137]. These manifestations are dose related. It can be controlled by either, reducing the total daily dose, dividing the total dose into multiple small doses, administrating the vitamin with food to decrease its absorption or to take the buffered form of vitamin C as sodium ascorbate or calcium ascorbate.
Advertisement
10. Contraindications of vitamin C administration
There are no contra-indications of vitamin C administration. Diabetics, patients prone to recurrent renal calculi, those undergoing stool occult blood tests, and those on sodium restricted diets or anticoagulant therapy should not take excessive doses of ascorbic acid over an extended period of time.
Diabetics taking more than 500 mg of ascorbic acid daily may obtain false readings of their urinary glucose test. No exogenous ascorbic acid should be ingested for 48–72 h before amine dependent stool occult blood tests are conducted because possible false-negative results may occur.
11. Vitamin C side effects
11.1 Oral route side effects
Side effects of vitamin C could be only detected with large doses exceeding the ULs for each individual especially on a single intake. Most of the vitamin C drawbacks were reported during oral uptake. They include diarrhea, abdominal pain [13], renal stones [123, 124] and enamel erosion during chewing [160].
11.1.1 GIT disturbance
Because of the poor oral bioavailability of vitamin C, toxic signs and symptoms may appear with large doses as a single dose. Diarrhea can occur. To avoid diarrhea occurrence, 2 g is the maximum permissible single dose [100]. Diarrhea and abdominal pain may occur due to the excretion of large amount of un-metabolized vitamin C [161]. Such manifestations are dose related. It can be controlled by either, reducing the total daily dose, dividing the total dose into multiple small doses, administrating the vitamin with food to decrease its absorption or to take the buffered form of vitamin C as sodium ascorbate or calcium ascorbate [100]. Even the usage of encapsulated vitamin C could not protect against the gastric upset. The gastrointestinal symptoms usually disappear within 1–2 weeks [19, 125].
11.1.2 Renal stones
Vitamin C metabolism results in calcium oxalate salts. Formation of renal stones (oxalate salts) and oxaluria are resulted in overdoses of vitamin C [162]. Later on, it was detected that this oxaluria is usually due to laboratory artifact occurs in the urine collection tube (ex vivo). It was also detected that vitamin C counteracting the formation of calcium oxalate crystals because of its ability to bind to calcium found in urine. Vitamin C also has the ability to increase the solubility of the calcium oxalate due to its mild acidity. It also triggers the normal urination process and prevents urine retention. All the previous actions decrease the incidence of kidney stones formation [123].
Also, it was found that vitamin C increases the urinary execration of uric acid and decreasing the plasma level of uric acid. But others demand the uricosuric effects of vitamin C as the rapid migration of uric acid from tissues. It was detected that 1 or 2 g per day increases the urinary oxalate stones were detected. Some studies detect lowering of urine pH after vitamin C intake [123].
The renal stones incidence of accumulation occurs in an average concentration of 60 g (IV) and more than 5 g (oral) [163]. In case of renal insufficiency, 1 g/day for 3 months is enough to produce renal stones (Alkhunaizi and Chan, 1996).
11.1.3 Metabolism side effects
It accelerates the absorption of other heavy metals as lead and mercury which increases its toxicity (Wyngaarded, 1987). In patients with high iron stores, vitamin C worsens the state. It also increases the iron-induced oxidative damage (Slivaka et al., 1986).
11.1.4 Dental side effects
Enamel erosions were detected on chewing of vitamin C tablets [126]. It was found that the usage of unbuffered form of ascorbic acid could result in enamel erosion [125].
11.2 Parenteral route side effects
During injection, transited mild soreness occurs during intramuscular, subcutaneous routes. Faintness or dizziness was reported on rapid intravenous administration. The renal stones incidence of accumulation occurs in an average concentration of 60 g (IV) and more than 5 g (oral) [163]. In case of renal insufficiency, 1 g/day for 3 months is enough to produce renal stones (Alkhunaizi and Chan, 1996).
12. Vitamin C deficiency
In scurvy, absence of wound healing and failure of fractured bones to heal. This was explained by the deficiency of collagen formation due to the vitamin C deficiency. Scurvy could be produced if reduction of the body reservoir of vitamin C into its fifth. The required body reservoir and the needed dosage are determinate according to the body weight [127].
In scurvy, body weakness, legs and arms edema, nose, skin and gums hemorrhage, infections, bone and cartilage damage (osteoporosis), vasculitis and cardiomegaly [164]. Many forms of bleeding found as petechiae, subperiosteal hemorrhage, ecchymoses, purpura, bleeding gums, hemarthrosis [165].
© 2018 The Author(s). Licensee IntechOpen. This chapter is distributed under the terms of the Creative Commons Attribution 3.0 License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
How to cite and reference
chapter statistics
2060 total chapter downloads
1 Crossref citations
More statistics for editors and authors
Login to your personal dashboard for more detailed statistics on your publications.
Access personal reporting
Related Content
This Book
Next chapter
Vitamin C: An Epigenetic Regulator
By Fadime Eryılmaz Pehlivan
Related Book
First chapter
Introductory Chapter: B-Group Vitamins
By Jean Guy LeBlanc
We are IntechOpen, the world's leading publisher of Open Access books. Built by scientists, for scientists. Our readership spans scientists, professors, researchers, librarians, and students, as well as business professionals. We share our knowledge and peer-reveiwed research papers with libraries, scientific and engineering societies, and also work with corporate R&D departments and government entities.
More About Us
Is Vitamin C Acidic Or Alkaline
Source: https://www.intechopen.com/chapters/64659


0 Komentar